|
Great Thanks to Tina Nye (A-5; 05-06) for much work on graphics: making a rough idea reality!
Chemical equilibrium
Calculation of K eq and Concentrations: After you have finished this you should be able to: 1) Calculate the equilibrium constant. 2) Calculate the equilibrium concentration of a participant when the value of K eq is known as well as the concentrations of the other participants. 3) Determination of the net direction of a reaction prior to establishing an equilibrium. You'd start from the expression for the change in Gibbs' free energy, DeltaG, relative to a reference, DeltaG^@, at standard pressure and a convenient temperature: DeltaG = DeltaG^@ + RTlnQ where: Q is the reaction quotient for the current state of the reaction. R and T are known from the ideal gas law. RTlnQ describes the shift in the free energy in reference to standard pressure and the. Keq equation calculator: calculate the equilibrium constant for the decomposition of water: how to calculate equilibrium constant for a reaction: calculate standard free energy change from equilibrium constant: calculate solubility product constant: calculate the cell potential the equilibrium constant and the free energy change for ba.
A system in equilibrium is like our ants up there! As long as the ants work at the same speed, the piles of sand remain in equilibrium. Neither gets bigger or smaller (It is important to note however that the piles are not the same size!). The work that each ant does exactly offsets what the other is doing. Products are being stacked up and the same rate that they are being taken away. Reactants are being stacked up at exactly the same rate that they are being taken away. If this were a chemical reaction, reactants would be colliding to make product at the same rate that products would be colliding with each other to make fresh reactant again! The reaction never really stops, but the forward reaction and reverse reaction proceed and the same rate, so we don't see a change in the size of the piles! Nifty Huh!!!!?
How To Calculate Keq Thermo
Chemical equilibrium is reached when the rates for the forward and reverse chemical reactions are equal for a chemical system. Or you could say when products are being made as fast as they are breaking down to form reactants again. The concentrations of products and reactants are generally NOT EQUAL.
To know what the concentrations are, you use the Equilibrium Constant expression (Keq).
Keq = [products]/[reactants]. Its a ratio : ) So Keq > 1 favors products, Keq < 1 favor reactants.
For a general equation like:
wA + xB <> yC + zD Remember, the square brackets mean concentration, in molarity, if it is given in some other unit you must calculate molarity. Coefficients in the chemical equation become exponents in the Keq expression. Leave out any solids or pure liquids since they have undefined molarities ; ).
Keq= If you spend time with the following notes, you'll master this topic in no time.
Predicting final concentrations and cheating while were doing it.
A chemical system can be thought of as being either: 1.At equilibrium 2.Not at equilibrium A system which is not at equilibrium will move spontaneously to a position of being at equilibrium. What is Keq : The 'K' in Keq stands for 'Constant'. The 'eq' means that the reaction is at equilibrium. Very roughly, Keq tells you the ratio of Products/Reactants for a given reaction at equilibrium at a certain temperature. K eq = [Products] / [Reactants] 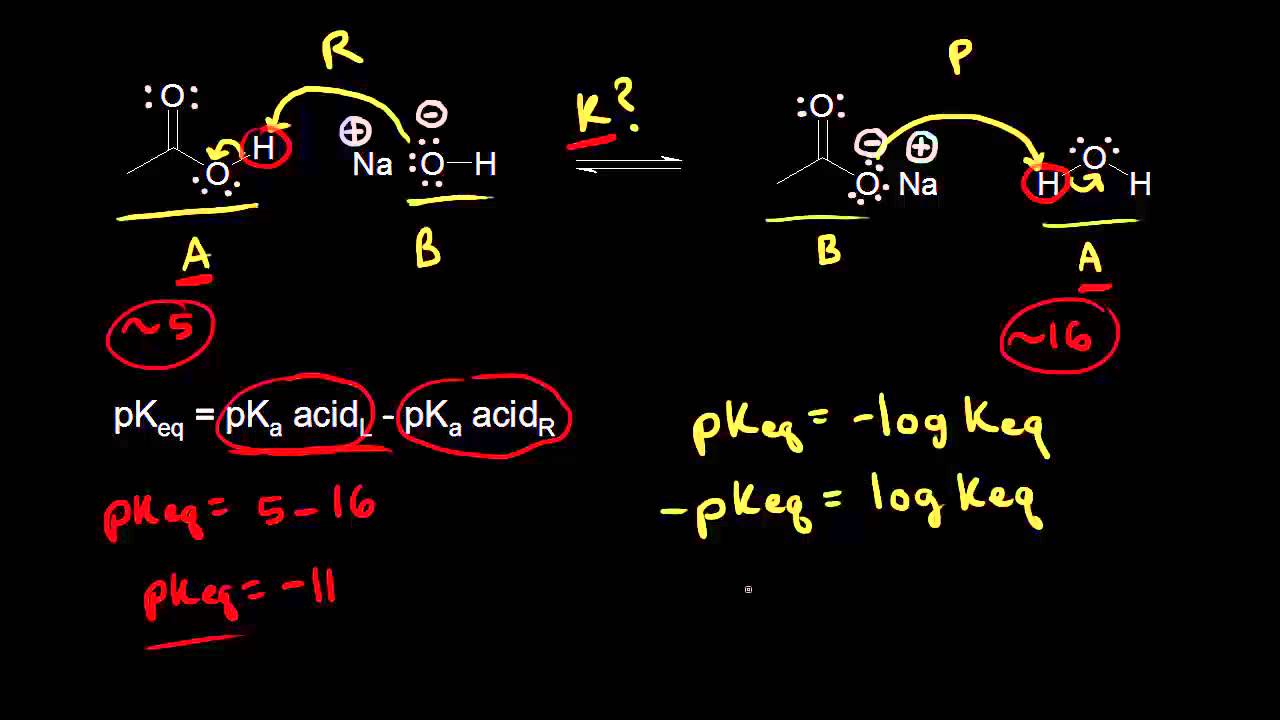 Consider the reaction
2HI(g ) < ---------------> H2(g) + I2(g)
that if you take the [H2], the [I2] and the [HI] in an equilibrium mixture of these at 423 °C,the expression [H 2 ] [I 2 ] / 2 [HI] = 0.0183
this is Keq expression this is value of Keq
Writing Keq Expressions:
Consider the general reaction:
aA + bB <------> cC + dD
where, A,B,C,D denote the reactants and prodcuts and a,b,c,d are coefficients in the balanced chemical equation.The equilibrium-constant expression for a reaction is an expression obtained by multiplying the concentrations of products,dividing by th concentration of reactants and raising each concentration term to a power equal to the coefficent in the chemical equation.
The equilibrium constant Kc is the value obtained for the equilibrium-constant expression when equilibrium concentrations are substituted.
Keq= [C]c [D]d / [A]a [B] b
Here you denote the molar concentration of a substance by writing its formula is square brackets.The subscript eq on the equilibrium contant means that it is defined in terms of molar concentrations.
The Keq Expressions for Solids and Liquids
How To Calculate Keq From Pka Values
For solids: When we write the Keq expression for a reaction with solids, we simply leave out the solids.
the Keq expression for the equation: CaCO3(s) <---------> CaO(s) яАл + CO2(g)
is simply: Keq = [CO2]
How To Calculate Keq With 0 VolumeThe same argument that was used for solids can also be used for liquids. Thus, we can expand the last statement: 'When we write the Keq expression for a reaction with solids or liquids, we simply leave out the solids and the liquids'. Gases and aqueous solutions do undergo changes in concentration so they are always How To Calculate Keq With Temperature
included in the Keq expression.
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Bitcommet
- Synthedit For Mac
- Sandisk Secure Access V2
- Driver Smart Modular Technologies Flash Card
- Kannada New Songs 2018 Download
- Idm Old Version With Crack Free Download
- Fonepaw Data Recovery 2 0 0 1
- Sony Dv Camera Driver For Mac
- Snapchat Macbook Air
- How To Create Email Templates In Outlook 2016 For Mac
- Vray For Mac
- Gta San Andreas For Pc Highly Compressed
- Videomark 1 7 – Easily Add Watermarks To Your Videos
- Turn Pc Into Mac Software
- Call Of Duty Modern Warfare 3 1.9.461
- Omnisphere 2 Response Code Generator
- Games Needed For Gmod
- Photoshop Software For Pc
- Rm 944 Usb Driver For Mac
- Iskysoft Imedia Converter Deluxe 5 4 0 Download Free
- Netplay For Mac
- Airfoil 5.6.0
- Mac Os X 10.5 Leopard Torrent Iso
- Sunn Amp Serial Number Lookup
- Pdf2id Free Mac
- Pro Tools Audio Editing Software Free Download
- Circular Studio 2 3
- 2pac Makaveli Album Tracklist
- Tomb Raider 3 Gold Cd Crack
- Anytime Organizer Torrent Download
- Isale 5 9 4 – Create And Post Ebay Auctions
- Gm Global Tis Keygen Software Download
- Jaksta Deluxe 2 0 0 Download Free
- George Of The Jungle 123
- Backstreet Boys Greatest Hits Zip
- Game Plants Vs Zombies 3 Full Version
- Free Fsx Iris Pro C27 Spartan Programs
- Badmash Hindi Rap Guru All Mp3 Songs Download
- 1991 Mercury Force 120 Hp Manual
- Adobe Premiere Pro Cs2 Free Full Version With Crack
 RSS Feed
RSS Feed
